2019 Geography Paper 1
SECTION A
Answer all the questions in this section.
1. (a) Distinguish between Geography and Environment.(2 marks)
(b) Identify the two branches of Geography.(2 marks)
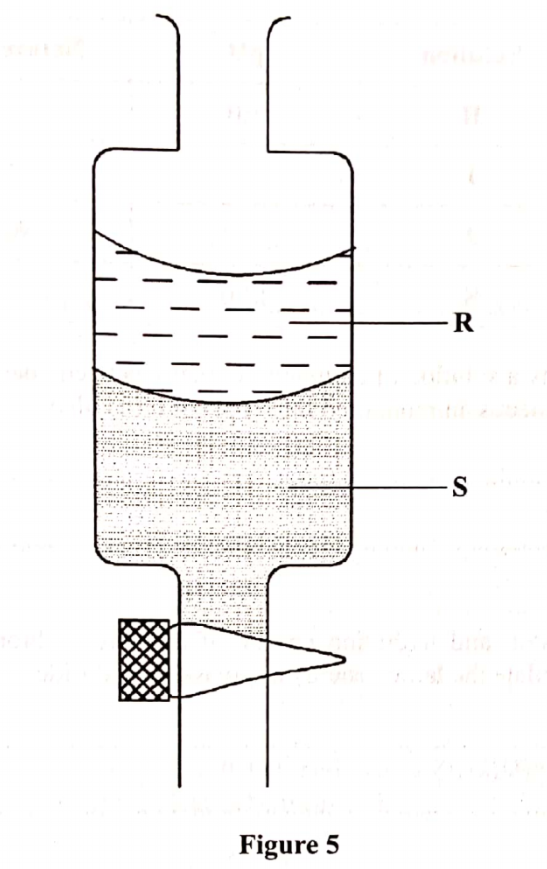
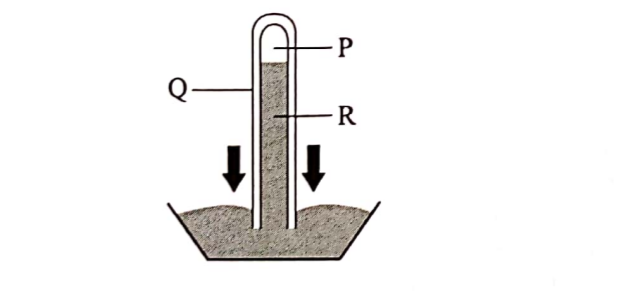
2. (a) The diagram below represents an instrument for measuring atmospheric pressure.
Name the parts marked P, Q and R.(3 marks)
(b) State three benefits of weather forecasting to human activities.(3 marks)
3. Give four proofs that support the theory of continental drift.(4 marks)
4. (a) Give three factors that influence the way a river transports its load.(3 marks)
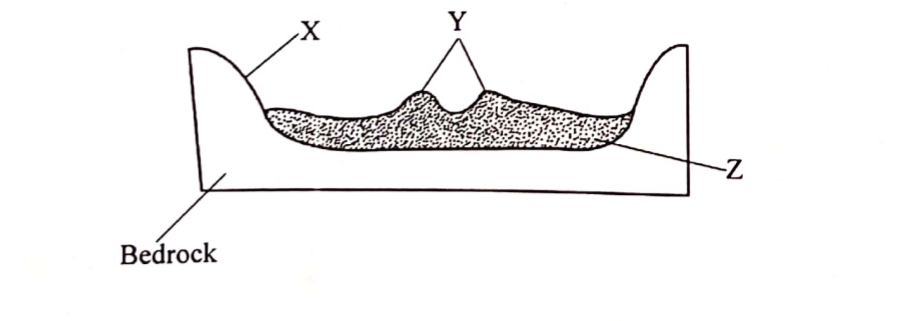
(b) The diagram below represents a cross-section of a flood plain. Name the features marked X, Y and Z. (3 marks)
5. (a) State the two causes of vertical movement of the ocean water.(2 marks)
(b) List three types of ocean tides.(3 marks)
SECTION B
Answer question 6 and any other two questions from fhis section.
6. Study the map of Yimbo 1: 50,000 (sheet l 15/1) provided and answer the following questions.
(â) (Î) What is the magnetic variation of the map?(2 marks)
(ii) Identify two natural features found at the grid square 3597.(2 marks)
(iii) Identify the two countries represented in the area covered by the map. (2 marks)
(b) (i) Give the direction of the trigonometrical station at the grid square 2789 from the air photo principal point at Nyagoma Mission School. (2 marks)
(ii) Measure the length of the provincial boundary to the North West of the area covered by the map. Give your answer in kilometres. (2 marks)
(c) (i) Give evidence that show the area covered by the map receives low rainfall. (3 marks)
(ii) Explain how each of the following factors have influenced the distribution of settlements in the area covered by the map:
I. Transport(2 marks)
II. Vegetation (2 marks)
III. Relief (2 marks)
(d) Describe the drainage of the area covered by the map.(6 marks)
7. (a) Using examples from East Africa, describe each of the following types of volcanoes:
(i) Active Volcano(3 marks)
(ii) Dormant Volcano(4 marks)
(b) Using a well labelled diagram, explain how each of the following features were formed:
(:) Composite Volcano (6 marks)
(ii) Volcanic plug (4 marks)
(c) Explain four negative effects of vulcanicity (8 marks)
8. (a) (i) Apart from inter tropical convergence zone (ITCZ), list four physical factors that influence climaie. (4 marks)
(ii) Give four charactcristics of inier tropical convergence zone (ITCZ). (4 marks)
(b) (i) Name the thrce equatorial climatic regions of Kenya. (3 marks)
(ii) Describe the tundra climate.(6 marks)
(c) Explain four human causes of desertification.(8 marks)
9. (a) (i) Define an ocean.(2 marks)
(ii) Explain the three causes of variation in the amount of salt in ocean water. * (6 marks)
(b) (i) Identify three processes involved in wave erosion. (3 marks)
(ii) With the aid of well labelled diagrams, describe the processes through which a wave cut platform is formed. (8 marks)
(c) You intend to carry out a field study on types of features resulting from wave deposition along the coast.
(i) Identify three methods you would use to record data. (3 marks)
(ii) Give three wave depositional features you are likely to observe during the field study.(3 marks)
10. (a) List three components of soil.(3 marks)
(b) Explain each of the following in relation to the classificati on of soil:
(i) Zonal(2 marks)
(ii) Intrazonal (2 marks)
(iii) Azonal (2 marks)
(c) Explain three causes of soil degeneration.(6 marks)
(d) Students from mwema school visited a nearby ranch to study types of soil.
(i) Explain why they carried the following tools:
i. Hoes (2 marks)
II. Polythene bags (2 marks)
(ii) Give three reasons why they would need to seek permission from relevant authorities. (3 marks)
(iii) List three types of soil erosion they are likely to observe. (3 marks)
Questions and Answers
2019 Geography Paper 1
SECTION A
Answer all the questions in this section.
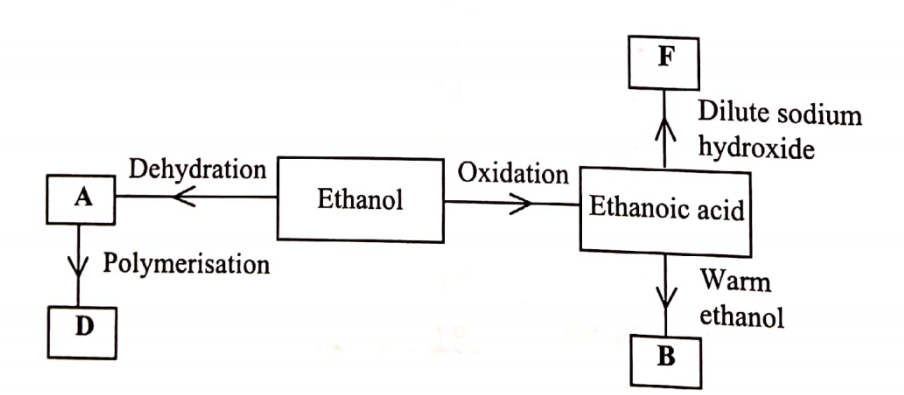
1.Distinguish between Geography and Environment
Geography refers to the study of the distribution of natural and human features/phenomenon and their interrelationship on the earth surface while Environment refers to external conditions that surround an organism and has influence on its behaviour.
(b) Identify the two branches of Geography.(2 marks)
– Physical Geography
– Huma Geography
2. (a) The diagram below represents an instrument for measuring atmospheric pressure.
Name the parts marked P, Q and R.(3 marks)
P – Vacuum
Q – Glass tube
R – Mercury

(b) State three benefits of weather forecasting to human activities.(3 marks)
– It enables farmers to plan their farming activities.
– It helps in guiding tourist aGtlVities.
– It enables military personnel to plan their military activities.
– It enables people to choose suitable clothing.
– It guides people on sporting activities.
– It guides people on fishing activities.
– It helps to determine the times for air/sea.
3. Give four proofs that support the theory of continental drift.(4 marks)
4. (a) Give three factors that influence the way a river transports its load.(3 marks)
(b) The diagram below represents a cross-section of a flood plain. Name the features marked X, Y and Z. (3 marks)

5. (a) State the two causes of vertical movement of the ocean water.(2 marks)
(b) List three types of ocean tides.(3 marks)
SECTION B
Answer question 6 and any other two questions from fhis section.
6. Study the map of Yimbo 1: 50,000 (sheet l 15/1) provided and answer the following questions.
(â) (Î) What is the magnetic variation of the map?(2 marks)
(ii) Identify two natural features found at the grid square 3597.(2 marks)
(iii) Identify the two countries represented in the area covered by the map. (2 marks)
(b) (i) Give the direction of the trigonometrical station at the grid square 2789 from the air photo principal point at Nyagoma Mission School. (2 marks)
(ii) Measure the length of the provincial boundary to the North West of the area covered by the map. Give your answer in kilometres. (2 marks)
(c) (i) Give evidence that show the area covered by the map receives low rainfall. (3 marks)
(ii) Explain how each of the following factors have influenced the distribution of settlements in the area covered by the map:
I. Transport(2 marks)
II. Vegetation (2 marks)
III. Relief (2 marks)
(d) Describe the drainage of the area covered by the map.(6 marks)
7. (a) Using examples from East Africa, describe each of the following types of volcanoes:
(i) Active Volcano(3 marks)
(ii) Dormant Volcano(4 marks)
(b) Using a well labelled diagram, explain how each of the following features were formed:
(:) Composite Volcano (6 marks)
(ii) Volcanic plug (4 marks)
(c) Explain four negative effects of vulcanicity (8 marks)
8. (a) (i) Apart from inter tropical convergence zone (ITCZ), list four physical factors that influence climaie. (4 marks)
(ii) Give four charactcristics of inier tropical convergence zone (ITCZ). (4 marks)
(b) (i) Name the thrce equatorial climatic regions of Kenya. (3 marks)
(ii) Describe the tundra climate.(6 marks)
(c) Explain four human causes of desertification.(8 marks)
9. (a) (i) Define an ocean.(2 marks)
(ii) Explain the three causes of variation in the amount of salt in ocean water. * (6 marks)
(b) (i) Identify three processes involved in wave erosion. (3 marks)
(ii) With the aid of well labelled diagrams, describe the processes through which a wave cut platform is formed. (8 marks)
(c) You intend to carry out a field study on types of features resulting from wave deposition along the coast.
(i) Identify three methods you would use to record data. (3 marks)
(ii) Give three wave depositional features you are likely to observe during the field study.(3 marks)
10. (a) List three components of soil.(3 marks)
(b) Explain each of the following in relation to the classificati on of soil:
(i) Zonal(2 marks)
(ii) Intrazonal (2 marks)
(iii) Azonal (2 marks)
(c) Explain three causes of soil degeneration.(6 marks)
(d) Students from mwema school visited a nearby ranch to study types of soil.
(i) Explain why they carried the following tools:
i. Hoes (2 marks)
II. Polythene bags (2 marks)
(ii) Give three reasons why they would need to seek permission from relevant authorities. (3 marks)
(iii) List three types of soil erosion they are likely to observe. (3 marks)