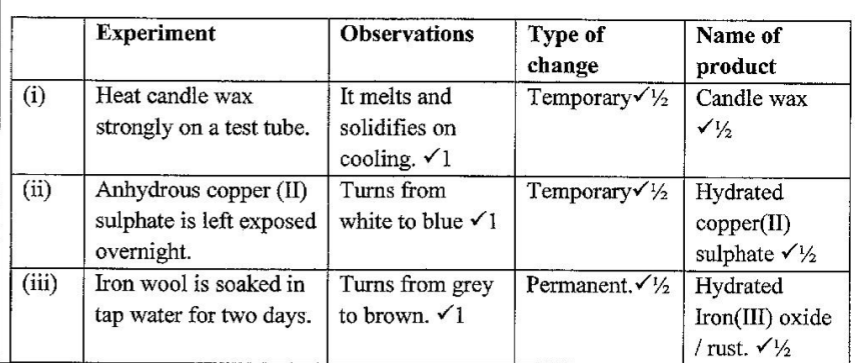
Kenya Certificate of Secondary Education
2018 Christian religious Education (CRE) Paper 1
1. (a)Outline seven teachings about human beings from the Biblical creation accounts. (7 marks)
(b) Give seven similarities between traditional African view of evil and the Biblical concept of sin. (7 marks)
2. (a)State six ways in which Christians in Kenya help to restore a member who has fallen into sin. (6 marks)
(b) Give five reasons why the Israelites broke the covenant they had made with God while at Mount Sinai (Exodus 32:1-35). (5 marks)
(b) Identify eight conditions that God gave to the Israelites during the renewal of the Sinai covenant (Exodus 34:1-35). (8 marks)
(c) State the importance of the Ten Commandments to Christians. (7 marks)
3. (a) Identify seven characteristics of the Canaanite religion. (7 marks)
(b) State the challenges faced by Elijah during his prophetic ministry in Israel. (6 marks)
(c) Give seven ways in which Christians fight comiption in Kenya today. (7 marks)
4. (a)Explain the vision of the basket of summer fruits shown to Prophet Arnos by God. (Amos 8:1-3). (6 marks)
(b) Outline the teaching of Prophet Amos on the day of the Lord. (7 marks)
(c) State seven ways in which Christians help the church leaders to perform their duties effectively. (7 marks)
5. (a) State the evils condemned by Prophet Jeremiah during the temple sermon. (6 marks)
(b) Explain four symbolic acts related to hope and restoration as demonstrated by Prophet Jeremiah. (8 marks)
(c)Give six lessons Christians learn from Prophet Jeremiah’s temple sermon. (6 marks)
6. (a)Outline six roles of priests in traditional African communities. (6 marks)
(b) Explain the importance of kinship ties in traditional African communities.( 5 marks)
(c) Identify six factors that have affected the kinship ties in the traditional African communities.(6 marks)
Questions and Answers
Christian Religious Education Paper 1
1. (a)Outline seven teachings about human beings from the Biblical creation accounts. (7 marks)
(i) Human beings are created in the image/likeliness of God
(ii) Human beings are co-creators with God/have to continue with God’s work of creation
(iii) Human life is God given/have the breath of life from God
(iv) They have the authority/dominion over God’s creation/superior to the rest of the creation.
(v) Human beings have the ability to thiñk/reason/make choices/decisions/bear consequence
(vi) Human beings are social beings/need companionship
(vii) They have a duty to take care of God’s creation/till the land/work
(viii) Human beings are to use other creation for their benefits/transform the world for the benefit
(ix) They are blessed by God
(x) Man and woman are equal before God
(xi) Human beings are to procreate through marriage/mutiply/fill the earth
(xii) They have been created to obey/fellowship with God
(xiii) The woman was created out of the man’s ribs.
(b) Give seven similarities between traditional African view of evil and the Biblical concept of sin. (7 marks)
(i) In both cases God/supreme being is good/not the author of sin/evil.
(ii) In both sin/evil is considered/known to interfere with the smooth running of the community as intended by God/supreme being.
(iii) In both sin/evil leads/results to alienation of human beings from God/interferes with good relationship.
(iv) Both sin/evil arises from human beings disobedience/greed/selfishness
(v) Both sin/evil causes a lot of suffering to human beings.
(vi) In both sin/evi1 may result from failing in a social/spiritual obligatiori/duty
(vii) In both human beings have the ability to overcome evil by choosing good/ right.
(viii) God is the guardian of morality/law/order in both.
(ix) In both sin/evil destroys the relationship among human beings/creates a situation of fear/mistrust/suspicion.
(x) In both sin/evil leads to punishment from God/can lead to destruction of God’s creation through natural calamities.
(xi) In both sin/evi1 leads to death
(xii) In both there is remedy for sin/evil
2. (a)State six ways in which Christians in Kenya help to restore a member who has fallen into sin. (6 marks)
(i) By praying for/with the member.
(ii) Through providing guidance and counselling/advice.
(iii) Visiting the member/doing a follow up after preaching to him/her.
(iv) By inviting him/her to come to church.
(v) Forgiving/accepting him/her in church.
(vi) Teaching/preaching the word of God to him/her.
(vii) By showing him/ her 1ove/ by providing material/financial support .
(viii) By encouraging him/her to confess/repent/seek for forgiveness
(b) Give five reasons why the Israelites broke the covenant they had made with God while at Mount Sinai (Exodus 32:1-35). (5 marks)
(i) Moses their leader had taken too long on the mountain.
(ii) Aaron, who had been left in charge of the peoples was weak/was easily manipulated.
(iii) The Israelites wanted a god they could see.
(iv) The Israelites were still influenced by the Egyptian way of worship/idolatry
(v) The teaching/influence of Moses had vanished from the people’s minds.
(vi) They lacked faith in God
(vii) They lacked knowledge of the nature of God.
(b) Identify eight conditions that God gave to the Israelites during the renewal of the Sinai covenant (Exodus 34:1-35). (8 marks)
(i) The Israelites were to obey God’s commands.
(ii) The Israelites were not to make anycovenant treaties with other tribes.
(iii) The Israelites were to destroy/break down the altars of their gods/smash their sacred stones/places of Idol worship.
(iv) They were not to worship any other god but Yahweh.
(v) They were to keep the feast of unleavened bread.
(vi) The Israelites were to dedicate all their first born male children/those of their flock to God.
(vii) The Israelites were to rest on the seventh day.
(viii) They were to offer to God the first fruits of their harvests/observe the feast of weeks.
(ix) All the male Israelites were to appear before the Lord God three times in a year.
(x) They were not to offer the blood of the sacrifices with leaven.
(xi) They were not to intermarry.
(xii) They were not to make graven images/idols.
(c) State the importance of the Ten Commandments to Christians. (7 marks)
(i) The Ten Commandments safegurds Christians against idol worship/promotes the worship of one true God.
(ii) They help Christians to have the right relationship with God/warns against indulging themselves in practices that drives them away from God/not to misuse God’s name.
(iii) They remind Christians to keep the Sabbath day/celebrate the Sabbath day by carrying out acts of charity/fellowshipping with one another.
(iv) They promote good relationship between parents and children/others.
(v) They help Christians to preserve/respect/protect human life.
(vi) They act as a guide to Christian living/safeguard marriages/bring stability in families.
(vii) The Ten Commandments help to protect pGople’s property/condemns all forms of theft.
(viii) They help to teach the virtue of honesty/self-control.
(ix) Ten Commandments promotes good relationship with other people/enhance peaceful co-existence.
3. (a) Identify seven characteristics of the Canaanite religion. (7 marks)
(i) Canaanite religion was a nature/cosmic/cyclic religion.
(ii) It comprised of many gods/goddesses/po1ytheistic.
(iii) They had a chief god/EL.
(iv) They worshipped their gods on mountain tops/high places of worship/temples/shrines.
(v) Symbols/idols/images were made to represent each god/goddesses.
(vi) The Canaanites practiced temple prostitution/was part of the worship of gods/goddesses.
(vii) They offered elaborate human/animal/crops sacrifices to the gods/goddesses.
L (viii) Feasts/festivals were celebrated in honor of gods/goddesses.
(ix) Each god had his/her own prophet/prophetess.
(x) Each god/goddess played a specific role in the community.
(b) State the challenges faced by Elijah during his prophetic ministry in Israel. (6 marks)
(i) Prophet Elijah had to defend his faith/religion to prove that Yahweh was the True God.
(ii) He was accused by King Ahab of being the trouble maker in Israel/enemy.
(iii) Prophet Elijah had to convince the people that he was the true prophet of Yahweh.
(iv) He lived in fear after pronouncing the drought in Israel.
(v) He had to flee from Israel after killing the prophets of Baal.
(vi) Jezebel threatened to kill him.
(vii) Elijah despaired/felt discouraged/lonely/wished to die.
(viii) True prophets of God were being persecuted/killed in Israel.
(ix) Elijah had to fight Baalism which had been made a state religion
(x) The presence of the many prophets of Baa1 in Israel
(c) Give seven ways in which Christians fight comiption in Kenya today. (7 marks)
(i) Christians fight corruption through condemning all the unjust practices in the society.
(ii) By praying for the corrupt people to change their behavior.
(iii) Through advocating for the enactment of laws that can help to strengthen the fight against comiption.
(iv) Christians act as role models for others to emulate/set good example by avoiding comipt practices.
(v) They make moral choices/informed decision/through the use of life skills. By lobbying for the punishment of all those who abuse justice/promote corruption,
(vi) By educating/providing civic education on the evils Of comiption.
(vii) Through exposing corrupt practices in the society.
(viii) By obeying the laws of the state.
(x) By preaching the importance of integrity to the people.
(ix) Christians repott those engaged in comtption to the relevant authorities
4. (a)Explain the vision of the basket of summer fruits shown to Prophet Arnos by God. (Amos 8:1-3). (6 marks)
(i) Prophet Amos was shown a basket of ripe fruits at the end of the harvest.
(ii) God told Amos that Israel was ripe for destruction.
(iii) Just as one waits for time to harvest fruits, God had waited for the Israelites to repent.
(iv) Since Israelites had not repented, the time had come for them to be punished.
(v) God’s judgement would not be delayed any longer.
(vi) Prophet Amos was silent/did not plead for God’s mercy for the Israelites.
(vii) This meant that punishment was inevitable.
(b) Outline the teaching of Prophet Amos on the day of the Lord. (7 marks)
(i) The day of the Lord would be full of darkness/no ligh/eclipse.
(ii) It would be a day of horror/disaster.
(iii) The day of the Lord will be a day of disappointment to the rich/rulers.
(iv) God would punish the (sraelites for their disobedience/He will remember their evil deed/God’s judgement would be upon them.
(v) There would be earthquakes/the land shall tremble/floods.
(vi) There will be mourning/wailing or the day of the Lord.
(vii) People will not delight in their feasts/festivals.
(viii) People will thirst/hunger for the word of God.
(ix) People will faint in the process of searching for the word of God.
(x) Israelites would be defeated by their enemies on the day of the Lord.
(xi) There would be famine/hunger/lack of food on the day of the Lord.
(xii) It will be a day of disappointment to the Israehtes/there will be no happiness/joy.
(c) State seven ways in which Christians help the church leaders to perform their duties effectively. (7 marks)
(i) Christians support church leaders financially/give material help/basic needs.
(ii) They defend them against unfair criticism.
(iii) By encouraging them iti their work/giving them moral support.
(iv) Advising/counseling them on various issues.
(v) By praying for/with them.
(vi) Respecting them/participate in church activities.
(vii) By practicing their teaching/obeying the word of God.
(viii) By giving tithes/offering to support the church activities.
5. (a) State the evils condemned by Prophet Jeremiah during the temple sermon. (6 marks)
(i) Prophet Jeremiah condemned the false belief that the temple could not be destroyed since it was God’s dwelling place.
(ii) He condemned the worship of other gods/idolatry.
(iii) He condemned the practice of human sacrifice which was against the commands of God.
(iv) He condemned hypocrisy among the people/syncretism/insincere worship.
(v) Prophet Jeremiah condemned the defilement of the temple/the people had put idols in it.
(vi) He condemned the oppression of the foreigners/widows/orphans which was against the covenant way of life.
(vii) He condemned the stubbornness of the Israelites against God’s warnings/had ignored the teachings of the prophets/failed to repent.
(b) Explain four symbolic acts related to hope and restoration as demonstrated by Prophet Jeremiah. (8 marks)
(i) The vision of two baskets of figs
Jeremiah saw a vision of two baskets of figs outside the temple. One basket had bad fruits that could not be eaten while the other one had good figs.
The good figs represented the first group who submitted to the deportation to exile.
This group of people would be restored/will be given new hearts/would be called people of God. The bad figs represented the group that refused to submit to the deportation to exile/would be punished.
(ii) The buying of land
God instructed Jeremiah to buy land from his cousin in Anathoth. He carried out the transaction before witnesses/gave the title deed to Baruch the scribe for safe keeping. This showed restoration of the Israelites to their homeland/They would reclaim the land.
(iii) The wooden Ox yoke
This symbolized the perseverance of the Jews in exile. After a period in exile, God would break the Babylonian yoke/restore the people back to the land/they would be set free.
(iv) The letter to the exiles
Jeremiah wrote the letter to encourage the exiles. They were to settle/build houses/have children/live in peace because they would return to Israel.
(v) Jeremiah’s visit to the potter’s house (c)Give six lessons Christians learn from Prophet Jeremiah’s temple sermon. (6 marks)
(i) Christians should have absolute trust/faith in God.
(ii) Christians should be morally upri%t/be exemplary.
(iii) They should warn people about judgement/punish the sinners/call sinners to repentance.
(iv) Christians should denounce/condemn social evils in the society.
(v) Christians should help the needy/widows/orphans.
(vi) They should be sincere in worship/shun hypocrisy/syncretism.
(vii) They should respect/ honour places of worship.
(viii) They should obey the word of God.
6. (a)Outline six roles of priests in traditional African communities. (6 marks)
(i) They give sacrifices to God/poor libations to ancestors.
(ii) They act as advisors of the community.
(iii) The priests intercede/offer prayers to God/spirits/ancestors on behalf of human beings.
(iv) They are the religious symbols of God’s presence in the community.
(v) In some communities the priest can act as a political leader.
(vi) They solve/settle disputes in the society/judge cases.
(vii) Priests preside over religious ceremonies/festivals.
(viii) They take care of shrines/places of worship.
(ix) They are custodians of customs/taboos/traditions of the community.
(b) Explain the importance of kinship ties in traditional African communities.( 5 marks)
(i) Kinship ties helps in preserving traditional customs through informal education.
(ii) They bind the entire life of a community/unites members of the same clari/family.
It helps to maintain law and order in the community.
Kinship ties promotes a sense of be1onging/security/identity. It defines the punishment/reward in the community.
It regulates marriage relationship in the community.
(vii) It helps in settling disputes in the community/family.
(viii) It defines duties/responsibilities of members of the clan/family.
(ix) It governs the relationship between the living and the dead.
(x) Kinship prepares the youth for leadership roles.
(xi) It enables people to share/help one another.
(c) Identify six factors that have affected the kinship ties in the traditional African communities.(6 marks)
(i) Formal education.
(ii) Modem religions/Christianity/Islam.
(iii) Mass media influence.
(iv) Permissiveness in the society.
(v) Western civilization.
(vi) Migration/individual land ownership.
(vii) New government structures/systems.
(viii) Economic factors/poverty/wealth/social status.
(ix) Urbanization.
(x) Modern science and technology.
kcse exam 2020
kcse 2020 English
kcse 2020 prediction
kcse 2020 mathematics prediction
kcse 2020 biology prediction
kcse 2020 biology paper 1
kcse 2020 timetable
kcse computer project 2020 question paper
kcse past papers
kcse past papers 2018
kcse past papers 2018 pdf download
FREE KCSE 2017 PAST PAPERS (QUESTIONS, MARKING SCHEMES AND REPORTS)
KCSE 2016 PAST PAPERS (QUESTIONS, MARKING SCHEMES AND REPORTS)
KCSE 2015 PAST PAPERS (QUESTIONS, MARKING SCHEMES AND REPORTS)
KCSE 2014 PAST PAPERS (QUESTIONS, MARKING SCHEMES AND REPORTS