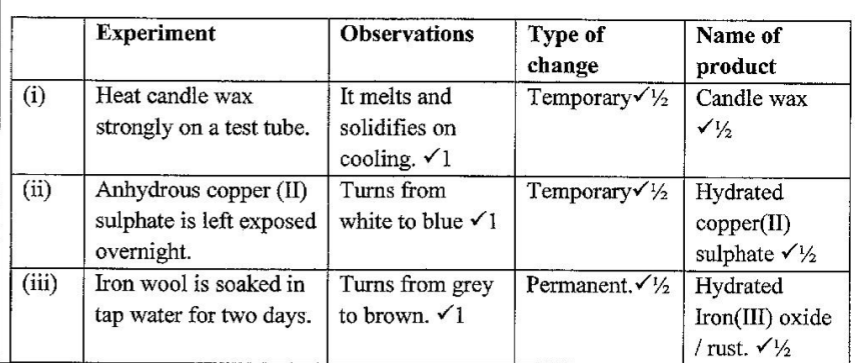
Kenya Certificate of Secondary Education
2018 Christian religious Education (CRE) Paper 2
1. (a) Outline Micah’s prophecy concerning the Messiah (Micah 5: 2-5).(7 marks)
(b) Describe the annunciation of the birth of Jesus by angel Gabriel as recorded in Luke I: 26-38.(8 marks)
(c) State five lessons Christians learn from the Magnificat. (5 marks)
2. (a) Explain the teachings of Jesus on the qualities of a true disciple (Luke 6: 20-49). (8 marks)
(b) Describe the incident in which Jesus foi’gave the sinful woman (Luke 7: 36-50). (7 marks)
(c) State five lessons Christians learn from the incident in which Jesus forgave the sinful woman. (5 marks)
3. (a) With reference to the story of the rich man and Lazarus, explain the teaching of Jesus on the proper use of wealth (Luke 16: 19-31). (7 marks)
(b) Outline six signs of the end times as taught by Jesus in Luke 21: 5-38. (6 marks)
(c) Give seven ways in which Christians are preparing themselves for the second coming of Jesus Christ. (7 marks)
4. (a) Identify eight characteristics of love as tau8ht by Saint Paul in I st Corinthians 13.(8 marks)
(b) Explain thc unity of believers as expressed in the concept of the church (Ephesians 5: 21-32). (5 marks)
(c) Identify seven factors that hinder effective cooperation among Christians in Kenya ioday. (7 marks)
5. (a) Outlinc the importance of professional Codes of Ethics. (6 marks)
(b) Identify seven factors that have led to an increased rate of child labour in Kenya loday. (7 marks)
(c) State seven ways through which Christ ians in Kenya are helping to reduce child labour in the society. (7 marks)
6. (a) Give seven reasons why Christians are opposed to plastic surgery. (7 marks)
(b) State seven effects of pollution on the environment in Kenya today. (7 marks)
(c) Identify six ways through which Christians in Kenya can conool desertification. (6 marks)
Questions and Answers
Christian Religious Education Paper 2
1. (a) Outline Micah’s prophecy concerning the Messiah (Micah 5: 2-5).(7 marks)
(i) The Messiah would be born in Bethlehem.
(ii) He will rule over Israel.
(iii) His origin is from the o1d/ancient days.
(iv) He would be born of a woman.
(v) He will feed His flock.
(vi) He will rule in majesty/strength of God.
(vii) In His time Israel will be secure/peaceful.
(viii) He shall be great to the ends of the earth/eternal.
(b) Describe the annunciation of the birth of Jesus by angel Gabriel as recorded in Luke I: 26-38.(8 marks)
(i) Angel Gabriel was sent/appeared to Mary in Nazareth.
(ii) Mary was a young woman who had been engaged to a man named Joseph from the house of David.
(iii) The angel greeted Mary “Hail, O favoured one, the Lord is with you.”
(iv) Mary was troubled by the greeting/wondered what it could be.
(v) The angel told her not to be afraid since she had found favour with God/was going to conceive/give birth to a son.
(vi) He would be called Jesus.
(vii) The angel told Mary that Jesus will be great/will be called the son of the Most High/God will give him the throne of his father David.
(viii) Mary asked the angel how she was going to conceive without a husband.
(ix) The angel told her that she would conceive by the power of the Holy Spirit.
(x) The angel told Mary that her kinswomen Elizabeth had also conceived a son/was six months pregnant
(xi) Mary accepted the angel’s message/said “I am the hand maid of the Lord, let it be to me as you have said.”
(xii) The angel departed from her.
(c) State five lessons Christians learn from the Magnificat. (5 marks)
(i) Christians should always appreciate/thank/glorify God for His favours.
(ii) Christians should have faith in God. (iii) Christians should be humble.
(iv) Christians should rejoice when they are blessed by God.
(v) Christians should obey/respect the will of God.
(vi) Christians should depend on God for provision.
2. (a) Explain the teachings of Jesus on the qualities of a true disciple (Luke 6: 20-49). (8 marks)
(i) Perseverance in the face of persecution — A true disciple should endure suffering.
(ii) Unwavering faith in Jesus — A true disciple should have absolute faith in God.
(iii) Love for enemies — A true disciple should love even their enemies/pray for them.
(iv) Obedience/implementers of the teachings of Jesus — A true disciple should accept the lordship of Christ/implement His teaching.
(v) Forgiving others — A true disciple should forgive others/not revenge.
(vi) Generosity/kindness — A true disciple should share with others/be generous.
(Vil) Not to judge others — A true disciple should exercise self-criticism before judging others.
(viii) Show compassion/mercy to others — A true disciple should be mercifiil just like God.
(b) Describe the incident in which Jesus foi’gave the sinful woman (Luke 7: 36-50). (7 marks)
(i) Simon the Pharisee invited Jesus to his house to eat with him.
(ii) A woman who was known to be a sinner came to Jesus with an alabaster flask of ointment.
(iii) She started seeping/wiped the feet of Jesus with her hair/kissed/anointed the feet of Jesus.
(iv) Simon wondered how Jesus could allow such a sinner to touch Him.
(v) Jesus knowing Simon’s thoughts decided to tell him the parable of the two debtors who owed their master different amounts of money and the master forgave them.
(vi) Jesus asked Simon who among the two debtors was more grateful.
(vii) Simon answered that the debtor who had more was more grateful.
(viii) Jesus told Simon that the sinful woman had shown a lot of love because her many sins had been forgiven.
(ix) Jesus told the woman to go in peace for her faith had saved her/her sins had been forgiven.
(c) State five lessons Christians learn from the incident in which Jesus forgave the sinful woman. (5 marks)
(i) Christians should confess/repent their sins.
(ii) The Kingdom of God is open to all/universal.
(iii) Christians should have faith in God.
(iv) Christians should be merciful/show compassion to those who are lost in sin.
(v) Christians should not judge others.
(vi) Christians should be grateful/thankful for the free gift of salvation/forgiveness of sins.
3. (a) With reference to the story of the rich man and Lazarus, explain the teaching of Jesus on the proper use of wealth (Luke 16: 19-31). (7 marks)
(i) Jesus taught that wealth should be used to alleviate the suffering of others/help the needy.
(ii) Wealth should be used to spread the good news.
(iii) Wealth should be used to serve others/glorify God.
(iv) Wealth should not be used to buy unnecessary materials for luxuries when others are suffering.
(v) Proper use of wealth enables one to be rewarded in heaven.
(vi) Wealth should not be used to oppress/humiliate the poor.
(vii) Too much trust in wealth can prevent someone from having eternal life/entering the kingdom of God.
(viii) Wealth is a gift/blessing from God/Christians are custodians of wealth.
(ix) Wealth is temporary/not permanent.
(b) Outline six signs of the end times as taught by Jesus in Luke 21: 5-38. (6 marks)
(i) Strange things will happen to the celestial bodies/sun/moon/star.
(ii) People will come claiming to be the Messiah.
(iii) There will be wars between nations.
(iv) Natural calamities like earthquakes/roaring of the seas will occur.
(v) The followers of Christ will be hated/persecuted.
(vi) There will be famine/pestilences.
(c) Give seven ways in which Christians are preparing themselves for the second coming of Jesus Christ. (7 marks)
(i) Christians pray/fast in readiness for the second coming of Jesus.
(ii) By accepting/confessing Jesus as their personal savior.
(iii) Living holy lives/pursuing righteousness.
(iv) Through preachlng the gospel to others/winning souls for Christ.
(v) Reading/practicing the word of God.
(vi) By fellowshipping/encouraging one another.
(vii) By helping the poor/needy.
(viii) Christians confess/repent their sins.
4. (a) Identify eight characteristics of love as tau8ht by Saint Paul in I st Corinthians 13.(8 marks)
(i) Love is patient.
(ii) It is kind.
(iii) It is not Jealous/boastful.
(iv) Love is not arrogant/rude.
(v) Love is not se1f-seeking does not insist on its own way.
(vi) Love is not irritable/resentful.
(vii) It does not rejoice in wrong/rejoices in the right.
(viii) Love bears all things.
(ix) Love endures/perseveres.
(x) Love believes/trusts all things.
(xi) Love never ends/it is eternal.
(xii) It is the greatest gift.
(b) Explain thc unity of believers as expressed in the concept of the church (Ephesians 5: 21-32). (5 marks)
(i) Paul used the symbol of marriage to show the unity of believers.
(ii) Christians are united to Christ just as a husband is united to his wife.
(iii) Christ loves the church just as the husband loves the wife.
(iv) The church has to submit to Christ just as the wife submits to her husband.
(v) Christ is the head of the church just as the husband is the head of the wife.
(vi) Just as a husband loves his own body/nourishes it, so Christ loves the church/nourlshes it.
(c) Identify seven factors that hinder effective cooperation among Christians in Kenya ioday. (7 marks)
(i) Greed for power/leadership wrangles.
(ii) Competition to win more converts/members.
(iii) Materialism/desire to have more.
(iv) Discrimination based on gender/tribe/education/race 1/social status/disability.
(v) Political interference.
(vi) Different interpretation of the Bible/doctrinal/denominational differences.
(vii) Emergence of cults/idol worship.
(viii) Lack of equity in resources/funds.
(ix) Poor infrastructure/language barrier/poor transport/communication facilities.
(x) Lack of money/inadequate funds.
5. (a) Outline the importance of professional Codes of Ethics. (6 marks)
(i) Professional code of ethics safeguard professionals against being compromised ln their work/upholds their integrity.
(ii) They give guidance on the relationship among professionals/those they serve.
(iii) They contain regu1ations/ terms of employment/promotion/remuneration/termination of services.
(iv) They promote se1f-discipline/accountability for the work done.
(v) They act as a measure of quality services/determine the expected levels of performance.
(vi) They enable the professionals to earn public trust/respect from people they serve/maintain public image of the profession.
(b) Identify seven factors that have led to an increased rate of child labour in Kenya loday. (7 marks)
(i) Poverty in the family has forced children to work.
(ii) They are cheap to contract/quest for cheap labour by employers.
(iii) Children can easily be employed/sacked/fired because they don’t have avenues to help them fight for proper terms/conditions.
(iv) Children have no skills/cannot negotiate for better terms of service/experience/knowledge.
(v) Greed for money by the child’s parent/guardian.
(vi) Death of parents/some children has been 1ef1 to fend for themselves.
(vii) Tiuancy from home/schools whereby some children abandon homes/schools due to peer pressure.
(viii) Gender discrimination in some communities/boys preferred/given priorities than girls.
(ix) Children are given too much responsibility at an early age/negligence/irresponsib1e parents.
(c) State seven ways through which Christ ians in Kenya are helping to reduce child labour in the society. (7 marks)
(i) Christians are helping to reduce child labour in the society by fighting for the rights of children/dignity of children.
(ii) By reporting those who employ children.
(iii) By advocating for stringent measures to curb child labour.
(iv) Teaching/sensitizing the society the evils of child labour.
(v) By condemning child labour/preaching against it.
(vi) Putting up homes for orphans/neglected children.
(vii) Through guidance and counselllng of parents on responsible parenthood.
(viii) Providing education to children from poor backgrounds.
(ix) By being role models/not practicing child labour.
6. (a) Reasons why Christians are opposed to plastic surgery.
(i) Plastic surgery takes the place of God/cha1lenges God as the creator/interferes with God’S lmage.
(ii) It shows lack of appreciation for oneself.
(iii) There are high risks involved/poor health/infections.
(iv) Plastic surgery is equated to idolatry/worship of the body.
(v) Plastic surgery promotes some vices/pride.
(vi) Plastic surgery can lead to death.
(vii) It can lower one’s self-esteem/stress/guilt.
(viii) It can lead to isolation/social stigma/rejection.
(b) Effects of pollution on the environment in Kenya today.
(i) Pollution has led to infections/diseases in human beings/animals.
(ii) It has led to global warming/result in climatic change/ozone layer.
(iii) Pollution has contributed to inconsistent rainfall patterns/drought/famine.
(iv) It has led to ecological/imbalance/migration/extinction of some animalsfiirds.
(v) It has led to death of human beings/animals/plants/birds.
(vi) It has led to human/animal conflicts.
(vii) It has damaged buildings/wastage of resources.
(viii) It has affected marine life/bio-diversity.
(c) Ways through which Christians in Kenya can control desertification.
(i) Christians can control desertification by protecting water catchment areas.
(ii) By exploiting/using alternative sources of energy/fuel.
(iii) Condemning/discouraging the distraction of forests.
(iv) Christians should participate in the planting of trees/afforestation/reforestation.
(v) By using better methods of farming.
(vi) Sensitizing/creating awareness of environmental conservation.
(vii) Christians should report those who destroy forests/environment to relevant authorities.
kcse exam 2020
kcse 2020 English
kcse 2020 prediction
kcse 2020 mathematics prediction
kcse 2020 biology prediction
kcse 2020 biology paper 1
kcse 2020 timetable
kcse computer project 2020 question paper
kcse past papers
kcse past papers 2018
kcse past papers 2018 pdf download
FREE KCSE 2017 PAST PAPERS (QUESTIONS, MARKING SCHEMES AND REPORTS)
KCSE 2016 PAST PAPERS (QUESTIONS, MARKING SCHEMES AND REPORTS)
KCSE 2015 PAST PAPERS (QUESTIONS, MARKING SCHEMES AND REPORTS)
KCSE 2014 PAST PAPERS (QUESTIONS, MARKING SCHEMES AND REPORTS